The content of this website is no longer being updated. For information on current assessment activities, please visit http://www.globalchange.gov/what-we-do/assessment
Calcium Carbonate Saturation in Ocean Surface Waters
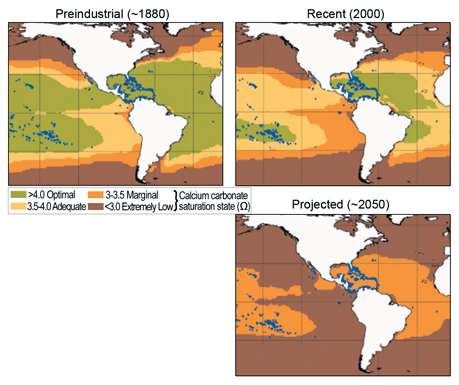
Corals require the right combination of temperature, light, and the presence of calcium carbonate (which they use to build their skeletons). As atmospheric carbon dioxide levels rise, some of the excess carbon dioxide dissolves into ocean water, reducing its calcium carbonate saturation. As the maps indicate, calcium carbonate saturation has already been reduced considerably from its pre-industrial level, and model projections suggest much greater reductions in the future. The blue dots indicate current coral reefs. Note that under projections for the future, it is very unlikely that calcium carbonate saturation levels will be adequate to support coral reefs in any U.S. waters.1 Image Reference: NAST1
References
- 1. a. b. [219] Team, National Assessment Synthesis. Climate Change impacts on the United States: The Potential Consequences of Climate Variability and Change. Cambridge, UK and New York, NY, 2001.
Summary
| High-resolution | High-resolution JPEG |
|---|